TheraPten Biosciences Inc.
Restoring functional PTEN tumor suppressor to stop cancer
Restoring functional PTEN to stop cancer
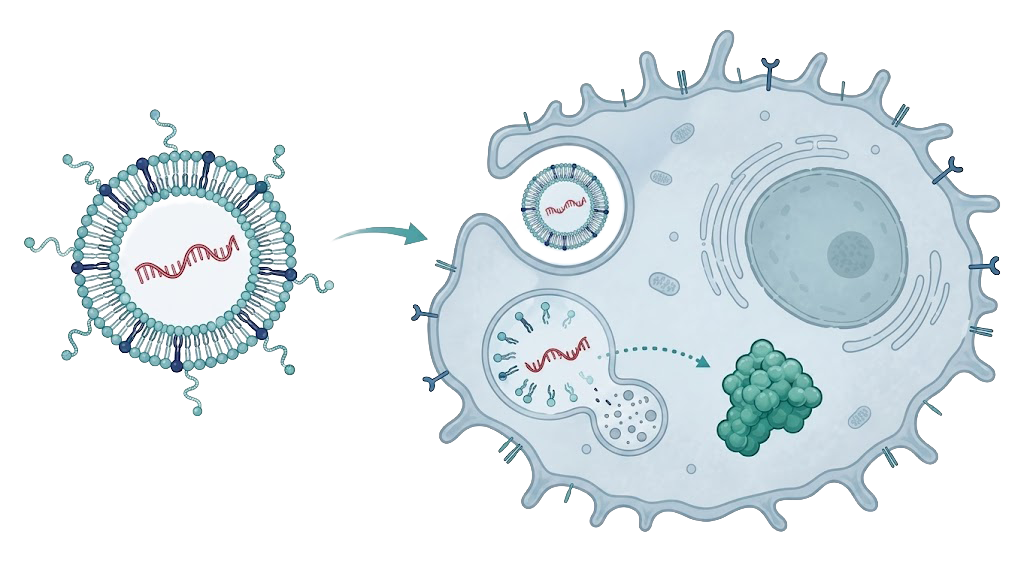
PR61 is designed to put functional PTEN back inside cancer cells — restoring a natural anti-cancer defense frequently lost across human tumors.
01
The therapy is designed to enter cancer cells directly and reset the body’s anti-cancer defense.
02
Clinical-trial-ready injectable therapy delivered using mRNA encapsulated in a lipid nanoparticle.
03
50–80% of cancer patients ultimately lose functional PTEN, regardless of tumor type or stage.
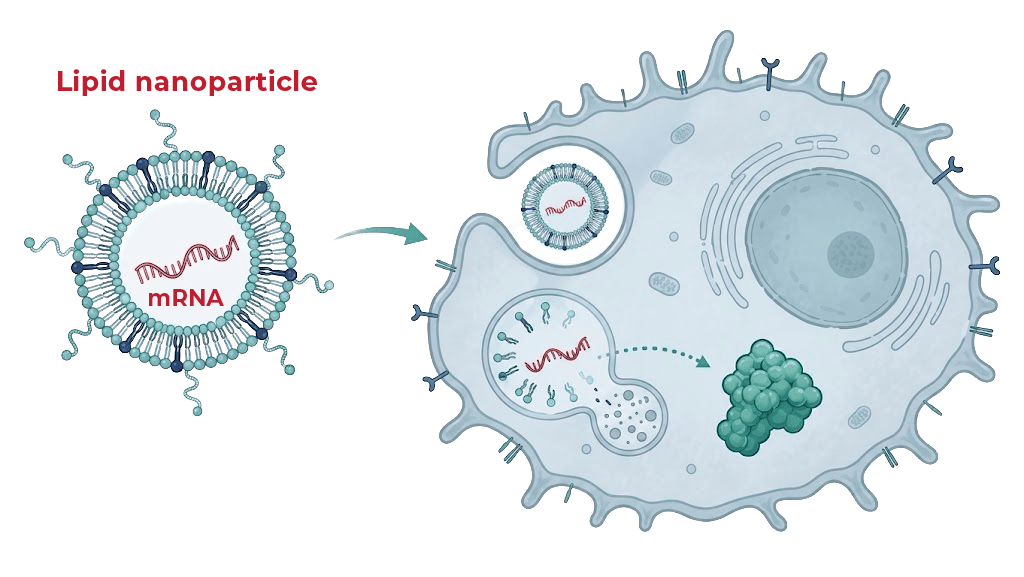
PR61 mRNA → lipid nanoparticle → intravenous injection
protein expression by the liver → functional PTEN expression


Drugging PTEN, an undruggable & elusive target
A cell-permeable PTEN approach designed to restore a naturally occurring tumor-suppressor pathway.
PTEN Biology
PR61 Profile
PR61 Mechanism of Action
Delivery and potential impact
Core invention
✓
PTEN is a natural human protein that suppresses tumors and promotes longevity.
✓
Loss of PTEN inside the cancer cell is one of the first events in cancer development.
✓
PTEN is missing or dysfunctional in 50–80% of human tumors across tumor type and stage.
✓
PTEN was identified in 1997 by Prof. Ramon Parsons at Columbia University; Prof. Parsons and Columbia University are TheraPten shareholders.
✓
Manufacturable to industrial standards
✓
Not toxic in preclinical studies
✓
Secreted from the human liver in large amounts
✓
Targets cancer cells- ✓Enters cancer cells directly to reset the body’s anti-cancer defense — the “Trojan horse” concept
- ✓ Approximately only 1% of roughly 35,000 natural and man-made proteins can enter cells directly.
- ✓ TheraPten has generated and patented PR61, a secretable & cell-permeable form of PTEN.
✓
PR61 is delivered by standard intravenous injection of PR61 mRNA encapsulated in a lipid nanoparticle, using the same broad delivery principle as mRNA vaccines.
✓
In animal studies, up to 100% of mice responded with significant tumor reduction and no toxicity.
✓
Up to 2 million cancer patients each year in the Western world alone could potentially benefit from treatment.
- ✓ Issued U.S. patent covering the PR61 albumin-PTEN fusion gene and protein.
- ✓ Patent applications pending in EU, China and U.S.
Initial oncology focus, with longevity as the next frontier of PTEN biology.
PR61 development pipeline
PR61 will initially be developed for two orphan cancer indications and one major cancer indication relevant to PTEN Hamartoma Tumor Syndrome (PHTS). PHTS patients are born with approximately 50% less PTEN and develop cancers with high propensity throughout life.
| Program | Indication focus | Rationale | Current positioning |
|---|---|---|---|
| PR61 Oncology | 2 orphan cancer indications | High-need settings where PTEN biology is central | Initial development focus |
| PR61 Oncology | 1 major cancer indication | Broader PTEN-deficient cancer opportunity | Expansion indication |
| PHTS-related cancers | PTEN Hamartoma Tumor Syndrome | Patients are born with ~50% less PTEN and have elevated lifetime cancer risk | Biology-driven rationale |
| PR61 Longevity | Healthy aging / longevity biology | PTEN overexpression studies show cancer resistance, cardiometabolic fitness benefits and 16% longer life expectancy vs controls | Next frontier / preclinical evidence |
PTEN replacement platform: Oncology first, then broader PTEN biology
TheraPten has obtained preclinical evidence that PR61 recapitulates at least one longevity aspect of PTEN.
The Expertise Behind TheraPten
Leadership & Advisors
Deep oncology, drug development, capital markets, and regulatory experience

Nassos Alevizopoulos, PhD, MBA
CEO & Chairman

Prof. Ramon Parsons, MD, PhD
Co-founder & Scientific Advisor

Maximilien Murone, PhD, MBA
Co-founder & Business Advisor

TheraPten Biosciences Inc.
Kelowna, British Columbia
Canada
reachout@therapten.com


